Introduction
Clinical resistance to the treatment of BCR-ABL1-positive chronic myeloid leukemia (CML) patients is often conferred by mutation(s) in the ATP-binding site of ABL1 kinase domain. ABL1 is subject to auto-inhibition mediated by myristoylation-triggered conformational change, which can be exploited to overcome resistance, as validated by allosteric inhibitors such as GNF2, GNF5 and asciminib. TGRX-678, a novel allosteric inhibitor designed to target the myristoyl pocket of ABL1, is currently being investigated in a phase I clinical trial (NCT05434312). Herein we report the selectivity, potency, anti-leukemia activity and in vivo pharmacokinetic (PK)/pharmacodynamic(PD) characteristics of TGRX-678.
Methods
For in vitro study, ABL1b 65-534 fragments were used following the protocol for Z'-LYTE kinase assay kit (Thermo Fisher). For cellular assays, CML cell lines or engineered Ba/F3 cells were incubated with titrating TGRX-678 for 72 hrs, then CellTiter Glo reagent (Promega) was added and the chemiluminescence was measured. For in vivo studies, SD rats received TGRX-678 or asciminib and plasma samples were collected for analysis. NOD-SCID mice were inoculated with CML or Ba/F3-BCR-ABL1 T315I cells and received oral TGRX-678. Tumor volumes and body weights were recorded for 3 to 5 weeks.
Results
TGRX-678 exhibited minimum off-target inhibition at 1 μM in a panel of 298 kinases, but inhibited the enzymatic activity of both ABL1 and the T315I mutant with IC50 values of 2.96 nM and 1.15 nM, respectively.
The anti-proliferative activity of TGRX-678 was determined in CML cell lines including K562, KU812, KCL22-S, KCL22-R, LAMA-84 and MEG-01 with IC50 values ranging from 1.1 nM to 6.56 nM. TGRX-678 had no cytotoxicity in 29 cell lines representing other hematological malignancies and solid tumors. Using engineered Ba/F3 cell lines, TGRX-678 was active against the native BCR-ABL1 (IC50 = 4.11 nM) and the clinically prominent mutants including G250E (IC50 = 2.49 nM), Q252H (IC50 = 2.38 nM), Y253H (IC50 = 5.6 nM), E255K (IC50 = 1.01 nM), E255V (IC50 = 2.73 nM) and T315I (IC50 = 66.1 nM). The cellular activity of TGRX-678 was attenuated by mutations in the myristoyl pocket such as A337V, P465S, V468F and I502L or mutations in the SH3-kinase domain interface including P223S and K294E, underpinning its allosteric mode of action.
In SD rats, oral administration of TGRX-678 at 20 mg/kg achieved a maximum plasma concentration (C max) of 2369 ng/mL, which was 1.3-fold higher than that of asciminib. The overall exposure (AUC last) of TGRX-678 (23,147 h·ng/mL) was also 1.7-fold higher than that of asciminib. The oral bioavailability of TGRX-678 was 62%. In xenograft-bearing mice, oral dosing of TGRX-678 at 1 mg/kg QD (for KU812) or 3 mg/kg QD (for K562) was sufficient to completely inhibit tumor growth. In the Ba/F3-BCR-ABL1 T315I xenograft mice, TGRX-678 induced dose-dependent tumor regression and reduced tumor volume by 67% at 45 mg/kg QD dose.
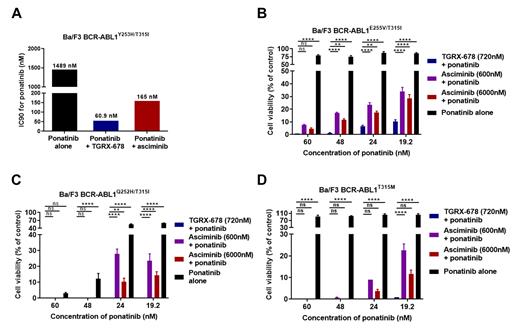
We further examined the synergistic effect of TGRX-678 and ponatinib combination. While each agent was inactive to the Y253H/T315I mutant, the combination of TGRX-678 and ponatinib inhibited cell proliferation at suboptimal concentrations. The addition of TGRX-678 dramatically lowered the IC90 for ponatinib from 1489 nM to 60.9 nM, whereas asciminib only reduced the IC90 to 165 nM (Figure A). Similarly, while ponatinib alone had limited effect on cell viability of the E255V/T315I, Q252H/T315I and T315M mutants, the addition of TGRX-678 at a clinically achievable concentration of 720 nM significantly reduced cell viability to 0-10 % (Figures B-D). In comparison, the addition of asciminib at a much higher concentration of 6000 nM only reduced cell viability to 5-29% (Figures B-D).
Conclusion
Our findings support that TGRX-678, a novel allosteric inhibitor of BCR-ABL1, is potent against the CML cells and cell lines harboring clinical ATP-site mutations with minimum non-specific cytotoxicity. TGRX-678 exhibits a favorable PK profile and anti-tumor activity in vivo. Furthermore, TGRX-678 and ponatinib synergize to overcome the clinically challenging resistance conferred by T315M or T315I-inclusive compound mutations. This data warrant further clinical investigation of TGRX-678 for the treatment of resistant or refractory CML patients.
Disclosures
Shi:Shenzhen TargetRx, Inc.: Current Employment, Current holder of stock options in a privately-held company. Li:Shenzhen TargetRx, Inc.: Current Employment, Current holder of stock options in a privately-held company. Jiang:Shenzhen TargetRx, Inc.: Current Employment, Current holder of stock options in a privately-held company. Yan:Shenzhen TargetRx, Inc.: Current Employment, Current holder of stock options in a privately-held company. Zheng:Shenzhen TargetRx, Inc.: Current Employment, Current holder of stock options in a privately-held company. Ai:Shenzhen TargetRx, Inc.: Current Employment, Current holder of stock options in a privately-held company. Wang:Shenzhen TargetRx, Inc.: Current Employment, Current equity holder in private company, Other: CEO of the company.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal